Molecular Biology (DENND1A)
Entry Clone Accession: BC039703
Entry Clone Source: MGC
SGC Construct ID: DENND1AA-c032
Protein Region: M1-N390
Vector: pFB-CT10HF-LIC
Tag: C-terminus flag tag preceded by a His10 tag. Both tags can be remove by TEV protease.
Host: DH10Bac
Sequence (with tag(s)): MGSRIKQNPETTFEVYVEVAYPRTGGTLSDPEVQRQFPEDYSDQEVLQTLTKFCFPFYVDSLTVSQVGQNFTFVLTDIDSKQRFGFCRLSSGAKSCFCILSYLPWFEVFYKLLNILADYTTKRQENQWNELLETLHKLPIPDPGVSVHLSVHSYFTVPDTRELPSIPENRNLTEYFVAVDVNNMLHLYASMLYERRILIICSKLSTLTACIHGSAAMLYPMYWQHVYIPVLPPHLLDYCCAPMPYLIGIHLSLMEKVRNMALDDVVILNVDTNTLETPFDDLQSLPNDVISSLKNRLKKVSTTTGDGVARAFLKAQAAFFGSYRNALKIEPEEPITFCEEAFVSHYRSGAMRQFLQNATQLQLFKQFIDGRLDLLNSGEGFSDVFEEEINAENLYFQSHHHHHHHHHHDYKDDDDK
Sequence after tag cleavage: MGSRIKQNPETTFEVYVEVAYPRTGGTLSDPEVQRQFPEDYSDQEVLQTLTKFCFPFYVDSLTVSQVGQNFTFVLTDIDSKQRFGFCRLSSGAKSCFCILSYLPWFEVFYKLLNILADYTTKRQENQWNELLETLHKLPIPDPGVSVHLSVHSYFTVPDTRELPSIPENRNLTEYFVAVDVNNMLHLYASMLYERRILIICSKLSTLTACIHGSAAMLYPMYWQHVYIPVLPPHLLDYCCAPMPYLIGIHLSLMEKVRNMALDDVVILNVDTNTLETPFDDLQSLPNDVISSLKNRLKKVSTTTGDGVARAFLKAQAAFFGSYRNALKIEPEEPITFCEEAFVSHYRSGAMRQFLQNATQLQLFKQFIDGRLDLLNSGEGFSDVFEEEINAENLYFQ
DNA Sequence: TTAAGAAGGAGATATACTATGGGCTCCAGGATCAAGCAGAATCCAGAGACCACATTTGAAGTATATGTTGAAGTGGCCTATCCCAGGACAGGTGGCACTCTTTCAGATCCTGAGGTGCAGAGGCAATTCCCGGAGGACTACAGTGACCAGGAAGTTCTACAGACTTTGACCAAGTTTTGTTTCCCCTTCTATGTGGACAGCCTCACAGTTAGCCAAGTTGGCCAGAACTTCACATTCGTGCTCACTGACATTGACAGCAAACAGAGATTCGGGTTCTGCCGCTTATCTTCAGGAGCGAAGAGCTGCTTCTGTATCTTAAGCTATCTCCCCTGGTTCGAGGTATTTTATAAGCTGCTTAACATCCTGGCAGATTACACGACAAAAAGACAGGAAAATCAGTGGAATGAGCTTCTTGAAACTCTGCACAAACTTCCCATCCCTGACCCAGGAGTGTCTGTCCATCTCAGCGTGCATTCTTATTTTACTGTGCCTGATACCAGAGAACTTCCCAGCATACCTGAGAATAGAAATCTGACAGAATATTTTGTGGCTGTGGATGTTAACAACATGTTGCATCTGTACGCCAGTATGCTGTACGAACGCCGGATACTCATCATTTGCAGCAAACTCAGCACTCTGACTGCCTGCATCCACGGGTCTGCGGCGATGCTCTACCCCATGTACTGGCAGCACGTGTACATCCCCGTGCTGCCGCCGCATCTGCTGGACTACTGCTGTGCTCCCATGCCCTACCTCATAGGAATCCATTTAAGTTTAATGGAGAAAGTCAGAAACATGGCCCTGGATGATGTCGTGATCCTGAATGTGGACACCAACACCCTGGAAACCCCCTTCGATGACCTCCAGAGCCTCCCAAACGACGTGATCTCTTCCCTGAAGAACAGGCTGAAAAAGGTCTCCACAACCACTGGGGATGGTGTGGCCAGAGCGTTCCTCAAGGCCCAGGCTGCTTTCTTCGGTAGCTACCGAAACGCTCTGAAAATCGAGCCGGAGGAGCCGATCACTTTCTGTGAGGAAGCCTTCGTGTCCCACTACCGCTCCGGAGCCATGAGGCAGTTCCTGCAGAACGCCACACAGCTGCAGCTCTTCAAGCAGTTTATTGATGGTCGATTAGATCTTCTCAATTCCGGCGAAGGTTTCAGTGATGTTTTTGAAGAGGAAATCAACGCAGAGAACCTCTACTTCCAATCGCACCATCATCACCATCACCATCACCACCATGATTACAAGGATGACGACGATAAGTGAGGATCC
Molecular Biology (RAB35A)
Entry Clone Accession: BC015931
Entry Clone Source: MGC
SGC Construct ID: RAB35A-c001
Protein Region: M1-C201
Vector: pNIC28-Bsa4
Tag: N-terminus His6 tag followed by a TEV cleavage site sequence
Host: BL21(DE3)-R3-pRARE2
Sequence (with tag(s)): MHHHHHHSSGVDLGTENLYFQSMARDYDHLFKLLIIGDSGVGKSSLLLRFADNTFSGSYITTIGVDFKIRTVEINGEKVKLQIWDTAGQERFRTITSTYYRGTHGVIVVYDVTSAESFVNVKRWLHEINQNCDDVCRILVGNKNDDPERKVVETEDAYKFAGQMGIQLFETSAKENVNVEEMFNCITELVLRAKKDNLAKQQQQQQNDVVKLTKNSKRKKRCC
Sequence after tag cleavage: SMARDYDHLFKLLIIGDSGVGKSSLLLRFADNTFSGSYITTIGVDFKIRTVEINGEKVKLQIWDTAGQERFRTITSTYYRGTHGVIVVYDVTSAESFVNVKRWLHEINQNCDDVCRILVGNKNDDPERKVVETEDAYKFAGQMGIQLFETSAKENVNVEEMFNCITELVLRAKKDNLAKQQQQQQNDVVKLTKNSKRKKRCC
DNA Sequence: CATATGCACCATCATCATCATCATTCTTCTGGTGTAGATCTGGGTACCGAGAACCTGTACTTCCAATCCATGGCCCGGGACTACGACCACCTCTTCAAGCTGCTCATCATCGGCGACAGCGGTGTGGGCAAGAGCAGTTTACTGTTGCGTTTTGCAGACAACACTTTCTCAGGCAGCTACATCACCACGATCGGAGTGGATTTCAAGATCCGGACCGTGGAGATCAACGGGGAGAAGGTGAAGCTGCAGATCTGGGACACAGCGGGGCAGGAGCGCTTCCGCACCATCACCTCCACGTATTATCGGGGGACCCACGGGGTCATTGTGGTTTACGACGTCACCAGTGCCGAGTCCTTTGTCAACGTCAAGCGGTGGCTTCACGAAATCAACCAGAACTGTGATGATGTGTGCCGAATATTAGTGGGTAATAAGAATGACGACCCTGAGCGGAAGGTGGTGGAGACGGAAGATGCCTACAAATTCGCCGGGCAGATGGGCATCCAGTTGTTCGAGACCAGCGCCAAGGAGAATGTCAACGTGGAAGAGATGTTCAACTGCATCACGGAGCTGGTCCTCCGAGCAAAGAAAGACAACCTGGCAAAACAGCAGCAGCAACAACAGAACGATGTGGTGAAGCTCACGAAGAACAGTAAACGAAAGAAACGCTGCTGCTGACAGTAAAGGTGGATACGGATCCGAA
Protein Expression (DENND1AA)
Medium: InsectXPRESS with L-Glutamine from Lonza
Antibiotics: none
Procedure: Sf9 cells were grown to 2x106 cells/mL, and infected with 3 mL of P2 virus. Cells were incubated at 27°C, shaking at 90 rpm for 48 h.
Protein Expression (RAB35A)
Medium: Autoinduction media
Antibiotics: Kanamycin and chloramphenicol.
Procedure: Each 1L culture was inoculated with 10 mL of a starter culture grown at 37°C overnight. When the OD600nm reached 0.5, the temperature was changed to 18°C and left
Protein Purification (DENND1AA)
Procedure: After expression, cultures were centrifuged at 1,000 g. After washing with PBS, pellets were frozen with liquid nitrogen. Pellets thawed and resuspended in lysis buffer (50 mM HEPES pH 7.5, 500 mM NaCl, 10 mM imidazole, 1 mM TCEP, 5% glycerol, 0.1% NP-40) containing Protease Inhibitors cocktail Set III from Calbiochem and Benzonase from Sigma. Cell disruption was carried out using a dounce homogenizer (10 passes with the loose pestle and 10 passes with the tight) followed by centrifugation at 35,000 g for 1h at 4°C. Lysates were applied to a 5 mL HisTrap column. The column was washed with buffer A (50 mM HEPES pH 7.5, 500 mM NaCl, 10 mM imidazole, 0.5 mM TCEP, 5% glycerol) followed by an additional wash containing 20% buffer B (50 mM HEPES pH 7.5, 500 mM NaCl, 250 mM imidazole, 0.5 mM TCEP, 5% glycerol). Final elution was carried out with 100% buffer B. The fractions containing DENND1A were dialyzed in buffer A at 4°C overnight, in the presence of TEV protease. The sample was diluted in a buffer containing 50 mM HEPES pH 7.5 and 100 mM NaCl and applied to a 1 mL HiTRAP Q capto column. Elution was performed by a linear gradient from 0 to 40% of a buffer containing 50 mM HEPES pH 7.5 and 1 M NaCl. The protein peak was concentrated and applied to a size exclusion chromatography HiLoad 16/600 superdex 200 pg column in a buffer containing 20 mM HEPES pH 7.0, 300 mM NaCl, 5% (v,v) glycerol and 0.5 mM TCEP. The peak corresponding with DENND1A was concentrated to 17 mg/mL. Purity was confirmed by SDS-PAGE.
Columns: Column 1: 5ml HisTrap; Column 2: TEV; Column 3: 1ml Q capto; Column 4: S200;
Concentration: 17.0 mg/mL
Compound Exact Mass: 466.014099
Protein Purification (RAB35A)
Procedure: After expression, cultures were centrifuged at 3,500 g and pellets were frozen. 10 g of bacterial pellet were resuspended in 100 mL of buffer A (20 mM HEPES pH 7.5, 200 mM NaCl, 0.5 mM TCEP) containing cOmplete EDTA-free protease inhibitors from Roche. Cell disruption was carried out using an Avestin EmulsiFlex-c5 homogenizer. Lysates were centrifuged at 35,000 g for 1h at 4°C. Samples were incubated with 2 mL of Ni-Sepharose 6 FF 1h at 4°C. After removing the non-bound lysate, beads were washed with buffer A followed by an extra wash with buffer A with 10 mM imidazole. Elution was achieved with buffer A containing 300 mM imidazole. Samples were then dialyzed into buffer A at 4°C overnight, in the presence of TEV protease. To keep the untagged BRD7, a second affinity chromatography using Ni-Sepharose 6 FF was performed. The flow through was then concentrated with an Amicon 3K centrifugal filter, and applied to a size exclusion chromatography column HiLoad 16/600 superdex 75 pg in a buffer A. The peak corresponding with RAB35 was concentrated to 15 mg/mL. Purity and protein identity were confirmed using SDS-PAGE, Q-TOF mass spectrometry, and tryptic digestion.
Columns: Column 1: 5ml HisTrap; Column 2: TEV cleavage; Column 3: Superdex 75 pg
Concentration: 15 mg/mL
Mass-spec Verification: Yes
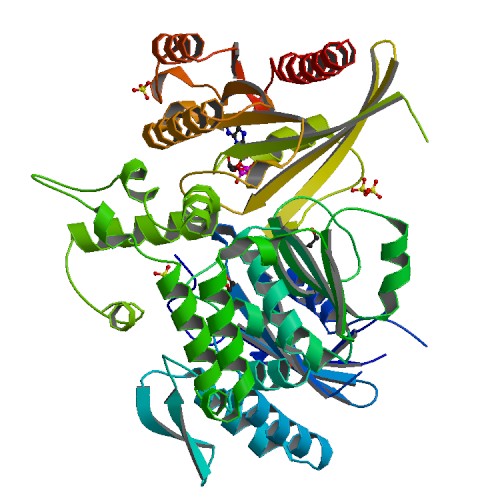
Structure Determination (DENND1AA:RAB35A)
Crystallization: Prior crystallization, DENND1A at 14 mg/mL was mixed with 15 mg/mL of RAB35 in a 1:1 volume ratio and incubated overnight at 4°C. Crystal plates were incubated at 4°C. Crystals grew in a condition containing 25% PEG3350, 0.2 M ammonium sulfate and 0.1M Bis-Tris pH 5.5. Crystals were cryo-protected with 15% ethylene glycol, and frozen in liquid nitrogen.
Data Collection: Diffraction data were collected at Diamond Light Source beamline I04-1 using a Pilatus 6M-F detector.
Data Processing: Data were indexed and integrated using Xia2, and scaled and analyzed with AIMLESS from the CCP4i suite. The structure was solved using MOLREP. The search model was the previously characterized DENND1A-RAB35 structure (PDB 3TW8). The resolution obtained was 1.82 Å. Refinement was carried out using PHENIX 1.9.