Molecular Biology
Entry Clone Accession:
Entry Clone Source: Collaborator - Georg Hollander
SGC Construct ID: FOXN1A-c017
Protein Region: P270-K366
Vector: pNIC28-Bsa4, N-terminal His tag followed by a TEV cleavage site.
Host: BL21(DE3)-R3-pRARE2
Sequence (with tag(s)): MHHHHHHSSGVDLGTENLYFQSMPKPIYSYSILIFMALKNSKTGSLPVSEIYNFMTEHFPYFKTAPDGWKNSVRHNLSLNKCFEKVENKSGSSSRKGCLWALNPAKIDKMQEELQKWKRK
Sequence after tag cleavage: SMPKPIYSYSILIFMALKNSKTGSLPVSEIYNFMTEHFPYFKTAPDGWKNSVRHNLSLNKCFEKVENKSGSSSRKGCLWALNPAKIDKMQEELQKWKRK
DNA Sequence: CATATGCACCATCATCATCATCATTCTTCTGGTGTAGATCTGGGTACCGAGAACCTGTACTTCCAATCCATGCCAAAACCCATCTATTCCTACAGCATCCTCATCTTCATGGCCCTTAAGAACAGTAAAACTGGGAGCCTTCCCGTCAGCGAGATCTACAATTTTATGACGGAGCACTTTCCTTACTTCAAGACAGCACCCGATGGCTGGAAGAATTCTGTCCGGCACAACCTATCCCTCAACAAGTGCTTCGAGAAGGTGGAGAACAAATCAGGAAGTTCCTCCCGCAAGGGCTGCCTGTGGGCCCTCAATCCGGCCAAGATCGACAAGATGCAAGAGGAGCTGCAAAAATGGAAGAGGAAATGACAGTAAAGGTGGATACGGATCCGAA
Protein Expression
Medium: Terrific Broth
Antibiotics: Kanamycin
Procedure: Recombinant protein expression was induced by the addition of 0.3 mM isopropyl 1-thio-- D-galactopyranoside to bacterial cultures grown in TB (Terrific Broth) containing 50 mg/ml kanamycin at an OD600 of 2.0 at 37 °C in UltraYield baffled flasks. Cultures were further incubated at 18 °C overnight.
Protein Purification
Procedure: Cell pellets were thawed and resuspended in buffer A (50 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol, 10 mM imidazole, 1 mM tris(2-carboxyethyl)phosphine (TCEP). Cells were lysed using sonication and cell debris was removed by centrifugation at 40,000 g for 1 h at 4 °C. Clarified lysates were applied to a 3-ml Ni-sepharose immobilized metal ion affinity chromatography gravity flow column, washed with 10 column volumes (CV) of buffer A, followed by 10 CV of wash buffer (buffer A with 45 mM imidazole). Proteins were eluted by addition of 5CV of buffer A containing 300 mM imidazole and analyzed by SDS-PAGE. Fractions containing FOXN1 were pooled and cleaved with the addition of His6-tagged TEV protease (1:20 mass ratio) overnight at 8 °C. Imidazole was removed by concurrent dialysis during cleavage, using a 3.5-kDa MWCO snakeskin membrane in buffer B (20 mM HEPES, pH 7.5, 500 mM NaCl, 5% glycerol, 0.5 mM TCEP). TEV protease was removed using a Ni-sepharose gravity flow column with cleaved FOXN1 being present in the flow through and 10 mM wash fractions. Cleaved protein was pooled and concentrated with a 3-kDa MWCO centrifugal concentrator. Final separation was by size exclusion chromatography, using a HiLoad 16/60 Superdex S75 column equilibrated in buffer B, and run at 1.2 ml/min.
Protein identity was confirmed by LC/ESI-TOF mass spectrometry with a single peak of mass 11505.4 corresponding to an expected mass of 11505.0.
Structure Determination
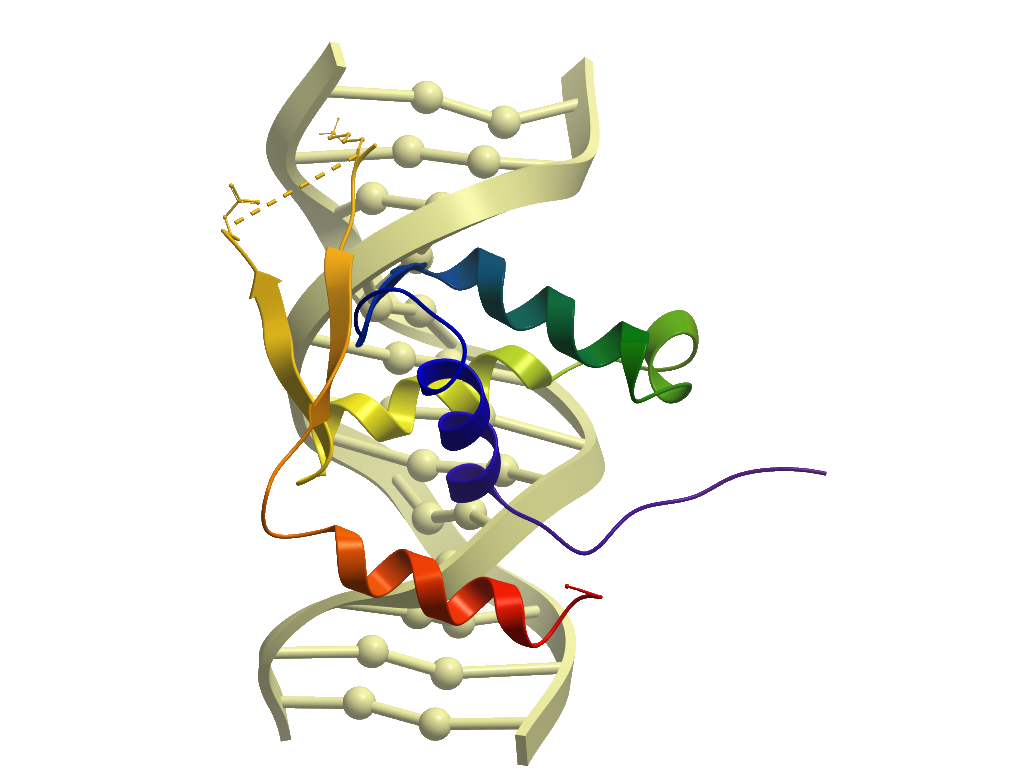
Crystallization: For crystallization the forkhead domain construct was concentrated to 10 mg/ml using a 10,000 mwco centrifugal concentrator and buffer exchanged to 10 mM Hepes pH 7.5, 250 mM NaCl, 0.5 mM TCEP. DNA was prepared by mixing the oligos 5’-GGTGGCGTCTTCA and 5’-TGAAGACGCCACC in a 1:1 ratio at a concentration of 500 µM, heating for 5 minutes at 94 °C, and letting cool slowly on a heat block. DNA and protein were mixed in a 1.2:1 molar ratio with final protein concentration of 5mg/ml. The FOXN1:DNA complex crystals were grown by vapor diffusion at 4°C in 150nl sitting drops with a 1:1 ratio of protein to precipitant. Crystals grew from conditions containing 8% PEG4000, 0.1M acetate pH 4.5. Crystals were cryo-protected by transferring to a solution of mother liquor supplemented with 25 % ethylene glycol and flash-cooled in liquid nitrogen.
Data Collection: Diffraction data were collected on beamline I04 at Diamond Light Source, at a wavelength of 0.975Å to a maximum resolution of 1.61Å. The data was processing using the program DIALS.
Data Processing: The structure was solved by molecular replacement using the program PHASER(20) with pdbID 26Y as a starting model. Model building and real space refinement were performed in COOT and the structure was refined using PHENIX REFINE to a final Rfactor=19.4 %, Rfree=23.9 %.