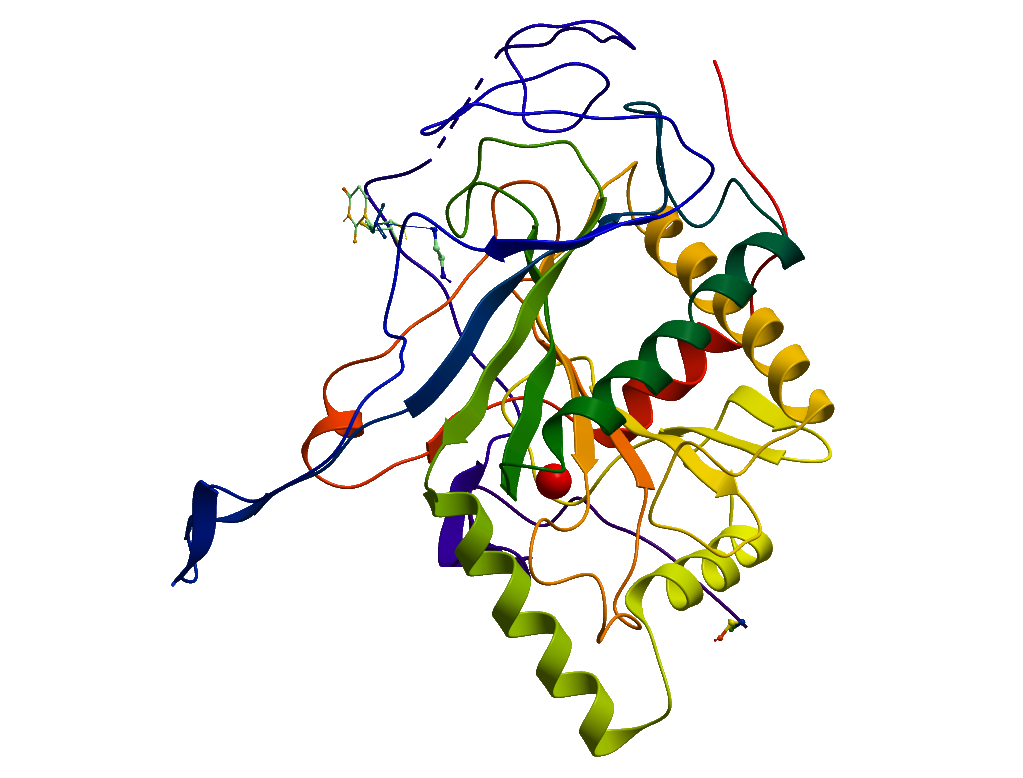
Molecular Biology
Entry Clone Accession: MGC:9490 IMAGE:3922902; note this clone harbours the p.Asn314Asp polymorphism
Entry Clone Source: MGC
SGC Construct ID: GALTA-c023 (A21Y:A22T:T23P:R25L mutation of GALTA-c001)
Protein Region: M1-A379
Vector: pNIC28-Bsa4
Tag: N-6HIS;N-TEV
Host: BL21(DE3)-R3-pRARE2
Sequence (with tag(s)): MHHHHHHSSGVDLGTENLYFQSMSRSGTDPQQRQQASEADAAYTPFLANDHQHIRYNPLQDEWVLVSAHRMKRPWQGQVEPQLLKTVPRHDPLNPLCPGAIRANGEVNPQYDSTFLFDNDFPALQPDAPSPGPSDHPLFQAKSARGVCKVMCFHPWSDVTLPLMSVPEIRAVVDAWASVTEELGAQYPWVQIFENKGAMMGCSNPHPHCQVWASSFLPDIAQREERSQQAYKSQHGEPLLMEYSRQELLRKERLVLTSEHWLVLVPFWATWPYQTLLLPRRHVRRLPELTPAERDDLASIMKKLLTKYDNLFETSFPYSMGWHGAPTGSEAGANWDHWQLHAHYYPPLLRSATVRKFMVGYEMLAQAQRDLTPEQAAERLRALPEVHYHLGQKDRETATIA
Sequence after tag cleavage: SMSRSGTDPQQRQQASEADAAYTPFLANDHQHIRYNPLQDEWVLVSAHRMKRPWQGQVEPQLLKTVPRHDPLNPLCPGAIRANGEVNPQYDSTFLFDNDFPALQPDAPSPGPSDHPLFQAKSARGVCKVMCFHPWSDVTLPLMSVPEIRAVVDAWASVTEELGAQYPWVQIFENKGAMMGCSNPHPHCQVWASSFLPDIAQREERSQQAYKSQHGEPLLMEYSRQELLRKERLVLTSEHWLVLVPFWATWPYQTLLLPRRHVRRLPELTPAERDDLASIMKKLLTKYDNLFETSFPYSMGWHGAPTGSEAGANWDHWQLHAHYYPPLLRSATVRKFMVGYEMLAQAQRDLTPEQAAERLRALPEVHYHLGQKDRETATIA
Note: this sequence represents a crystal epitope variant (A21Y:A22T:T23P:R25L) of the GALT protein
DNA Sequence: ATGCACCATCATCATCATCATTCTTCTGGTGTAGATCTGGGTACCGAGAACCTGTACTTCCAATCCATGTCGCGCAGTGGAACCGATCCTCAGCAACGCCAGCAGGCGTCAGAGGCGGACGCCGCATACACCCCTTTCCTGGCAAACGACCATCAGCATATCCGCTACAACCCGCTGCAGGATGAGTGGGTGCTGGTGTCAGCTCACCGCATGAAGCGGCCCTGGCAGGGTCAAGTGGAGCCCCAGCTTCTGAAGACAGTGCCCCGCCATGACCCTCTCAACCCTCTGTGTCCTGGGGCCATCCGAGCCAACGGAGAGGTGAATCCCCAGTACGATAGCACCTTCCTGTTTGACAACGACTTCCCAGCTCTGCAGCCTGATGCCCCCAGTCCAGGACCCAGTGATCATCCCCTTTTCCAAGCAAAGTCTGCTCGAGGAGTCTGTAAGGTCATGTGCTTCCACCCCTGGTCGGATGTAACGCTGCCACTCATGTCGGTCCCTGAGATCCGGGCTGTTGTTGATGCATGGGCCTCAGTCACAGAGGAGCTGGGTGCCCAGTACCCTTGGGTGCAGATCTTTGAAAACAAAGGTGCCATGATGGGCTGTTCTAACCCCCACCCCCACTGCCAGGTATGGGCCAGCAGTTTCCTGCCAGATATTGCCCAGCGTGAGGAGCGATCTCAGCAGGCCTATAAGAGTCAGCATGGAGAGCCCCTGCTAATGGAGTACAGCCGCCAGGAGCTACTCAGGAAGGAACGTCTGGTCCTAACCAGTGAGCACTGGTTAGTACTGGTCCCCTTCTGGGCAACATGGCCCTACCAGACACTGCTGCTGCCCCGTCGGCATGTGCGGCGGCTACCTGAGCTGACCCCTGCTGAGCGTGATGATCTAGCCTCCATCATGAAGAAGCTCTTGACCAAGTATGACAACCTCTTTGAGACGTCCTTTCCCTACTCCATGGGCTGGCATGGGGCTCCCACAGGATCAGAGGCTGGGGCCAACTGGGACCATTGGCAGCTGCACGCTCATTACTACCCTCCGCTCCTGCGCTCTGCCACTGTCCGGAAATTCATGGTTGGCTACGAAATGCTTGCTCAGGCTCAGAGGGACCTCACCCCTGAGCAGGCTGCAGAGAGACTAAGGGCACTTCCTGAGGTTCATTACCACCTGGGGCAGAAGGACAGGGAGACAGCAACCATCGCCTGA
Protein Expression
Medium: Auto induction Terrific Broth (50 g ForMedium AIM-TB, 20 g glycerol and water to 1 L) was autoclaved. After autoclaving 1 mL of 10 % Antifoam 204 (in ethanol) was added.
Antibiotics: Kanamycin (50 µg/mL) and Chloramphenicol (35 µg/mL)
Procedure: A 10 mL LB o/n culture was grown and used to inoculate 1 L of AIM-TB in a 2.5 L Ultra Yield baffled flask. Cells were grown for 4 h at 37 °C 250 rpm shaking and then for a further 40 h at 18 °C. Cells were harvested by centrifugation at 4,000 g for 20 minutes.
Protein Purification
Procedure: 3 mL of Base Buffer was added per g of cell pellet (10 mM HEPES, 500 mM NaCl, 5 % Glycerol, 0.5 mM TCEP, pH 7.5) + 0.5 mg/mL lysozyme, 1 µg/mL benzonase, 1 % Triton X-100, 30 mM imidazole. After stirring for 30 minutes at room temperature cells were frozen at -80 °C for 1-2 h. Cells were thawed at RT for 30 minutes and subsequently centrifuged at 4,000 g for 1 h at 4 °C. 40 mL of SN was then applied to a 1 mL His GraviTrap column (GE healthcare) fitted with a LabMate extender. The column was then washed twice with 10 mL of Base Buffer + 30 mM imidazole. The His GraviTrap column was then slotted into a PD-10 column fitted with a LabMate extender and the protein eluted with 2.5 mL of Base Buffer + 500 mM imidazole. The His GraviTrap columne was then removed and protein eluted from the PD-10 with 3.5 mL of Base Buffer + 30 mM imidazole. 1 part TEV was then added to 10 parts GALTA and the sample incubated o/n at 4 °C. . Cleaved protein was then run back over the His GraviTrap column to remove TEV and any un-cleaved protein. Protein was then concentrated to 20-40 mg/mL and run on a Bio-Rad SEC 650 gel filtration column using Base Buffer as the mobile phase. Peak fractions were pooled and concentrated to 15.1 mg/mL.
Concentration: 15.1 mg/ml
Mass-spec Verification: Yes, Expected 43526.5 Da, Observed 43526.1
Structure Determination
Data Collection: X-ray collection (SAD); Beamline: Dmnd I04-1; Resolution: 1.52 Å
Crystallization and Data Processing: Crystals were obtained after 2 weeks at 4 oC in reservoir solution containing 0.2M ammonium sulphate and 30% PEG4000. Crystals were cryo-protected with reservoir solution supplemented with 25% (v/v) ethylene glycol and flash-cooled in liquid nitrogen. The structures were solved by molecular replacement with PHASER, using the structure of E. coli GALT (1HXP) as template. Modelling and refinement were carried out using Refmac and Coot.