= Title =
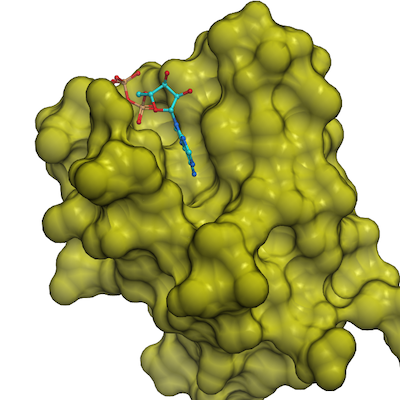
Crystal Structure of Human HUWE1 WWE domain in complex with ADPR
= Keywords =
HUWE1, WWE-domain, Transferase
= PDB Code =
6PFL
= Ligands =
APR, UNX
= Entry clone accession =
= Entry clone source =
= SGC clone accession =
YTC050-G10
= Tag =
N-terminal His6-tag with TEV cleavage site
= Construct comments =
= Construct sequence =
mhhhhhhssgrenlyfqgQSNSNNWRWFDDRSGRWCSYSASNNSTIDSAWKSGETSVRFTAGRRRYTVQFTTMVQVNEETGNRRPVMLTLLRVPRLNKNSKNSNGQEL
DNA sequence has been verified by sequencing
= Vector =
pET28-MHL
= Expression host =
BL21(DE3)V2R-pRARE2
= Growth method =
LEX system
The target protein was over-expressed in E. coli at 37°C by inoculating 20 mL of overnight culture grown in Luria-Bertani medium with 40ug/ml kanamycin and 25ug/ml chloramphenicol into 2L Terrific Broth medium in the presence of 40 ug/mL kanamycin. When the OD600 of the culture reached ~1.5, the temperature was lowered to 15 degree and the culture was induced with 0.5 mM IPTG. The cells were allowed to grow overnight before harvested by centrifugation (7,000 rpm Beckman JLA-8.1000 rotor 10 min) and flash frozen in liquid nitrogen and stored at -80°C.
= Extraction buffers =
20 mM Tris pH 8.0, 500 mM NaCl, 5% Glycerol
= Extraction procedure =
Native cell pellet was resuspended in extraction buffer and the cells disrupted by sonication for 10 mins at 5" on 10" off duty cycle at 90W output power.
= Purification buffers =
Washing Buffer: 20 mM Tris pH 8.0, 250 mM NaCl, 5% Glycerol, 50 mM imidazole
Elution Buffer: 20 mM Tris pH8.0, 250 mM NaCl with 5% glycerol, 250 mM imidazole
= Purification procedure =
The crude extract was cleared by centrifugation. The lysate was loaded onto Ni Sepharose FF column charged with Ni2+. The column was washed with 10 CV of 20 mM Tris-HCl buffer, pH 8.0, containing 250 mM NaCl, 50 mM imidazole and 5% glycerol, and the protein was eluted with elution buffer (20 mM Tris-HCl, pH 8.0, 250 mM NaCl, 250 mM imidazole, 5% glycerol).
The protein was further purified to homogeneity by Superdex200 column (26x60) (Amersham Biosciences), equilibrated with 20 mM PIPES buffer, pH 6.5, and 250 mM NaCl, at flow rate 4 ml/min.
TEV protease was added to combined fractions containing WWE doamin protein and incubated overnight at 4°C.
The protein was loaded onto ion-exchange chromatography on Source 30Scolumn, equilibrated with buffer 20mM Pipes pH6.5, and eluted with linear gradient of NaCl up to 1 M concentration (20CV).
The protein was final buffer exchanged to 20mM Tris [8.0], 150 mM NaCl.
Protein yield: 1.9 mg/L
= Protein stock concentration =
Concentration used for crystallization : 20 mg/mL
= Mass spec =
N/A
= Functional multimerization =
Monomer
= Crystallization =
Crystallization of the HUWE1-WWE domain in complex with ADP-ribose was performed in 96-well vapor diffusion sitting drop plates by mixing 1:10 ratio of WWE protein and ADPR and set plates using equal volumes of protein and reservoir solution at room temperature.
Diffraction quality crystals were obtained in 1.4 M Sodium Citrate, and 0.1 M TRIS-HCl [pH 7.5].
Crystals were cryoprotected in reservoir solution supplemented with 20% glycerol and then with paratone, and cryo-cooled in liquid-nitrogen.